Armoring T Cells to Eradicate CAR-Resistant Malignancies and Restore Immune Balance in Autoimmune Diseases
PrecisionAxis Bio is a pre-clinical stage biotechnology company pioneering next-generation 'Armored' cell therapies to overcome the primary challenges in the treatment of solid tumors and autoimmune diseases. By integrating proprietary, stabilized membrane-bound or soluble Tumor Necrosis Factor-related Apoptosis-Inducing Ligand (TRAIL) fusion proteins with Chimeric Antigen Receptor (CAR) technology, we eliminate tumor escape caused by antigen heterogeneity or antigen loss.
This dual-action mechanism triggers both antigen-dependent tumor cell lysis and potent, antigen-independent bystander apoptosis. Our approach is empowered by a 100-fold increase in TRAIL potency compared to clinical-grade TRAIL, ensuring:
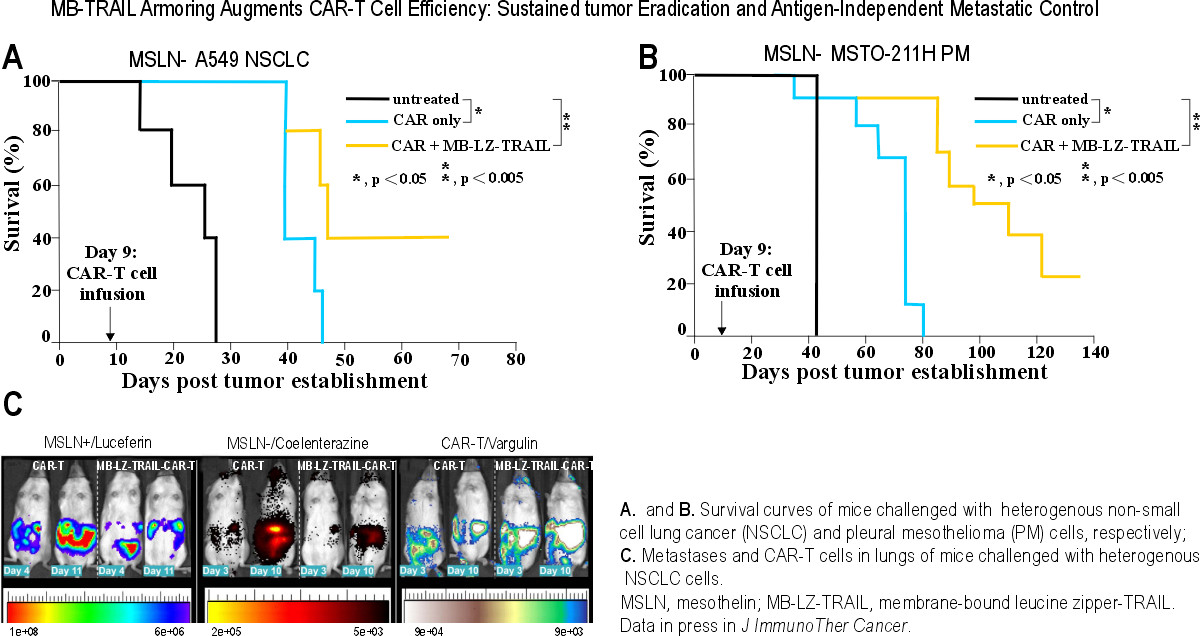
Durable Complete Responses: Achieving sustained tumor eradication in 40% and 20% of mice challenged with heterogeneous non-small cell lung cancer (NSCLC) and pleural mesothelioma (PM) tumor cells, respectively, where standard therapies fail.
Eradication of Metastatic Burden: Proven homing and potent killing of antigen-negative distant metastases, ensuring a comprehensive therapeutic effect.
Significant Survival Extension: Substantially increasing the therapeutic window and delaying progression in an aggressive tumor microenvironment (TME).
While our primary focus is complementing CAR-mediated treatment with the second, antigen-independent apoptotic pathway, our modular platform is also being engineered to reset immune balance in autoimmune pathologies, leveraging the same precision-targeting principles to protect healthy tissue and eliminate pathogenic immune cells.
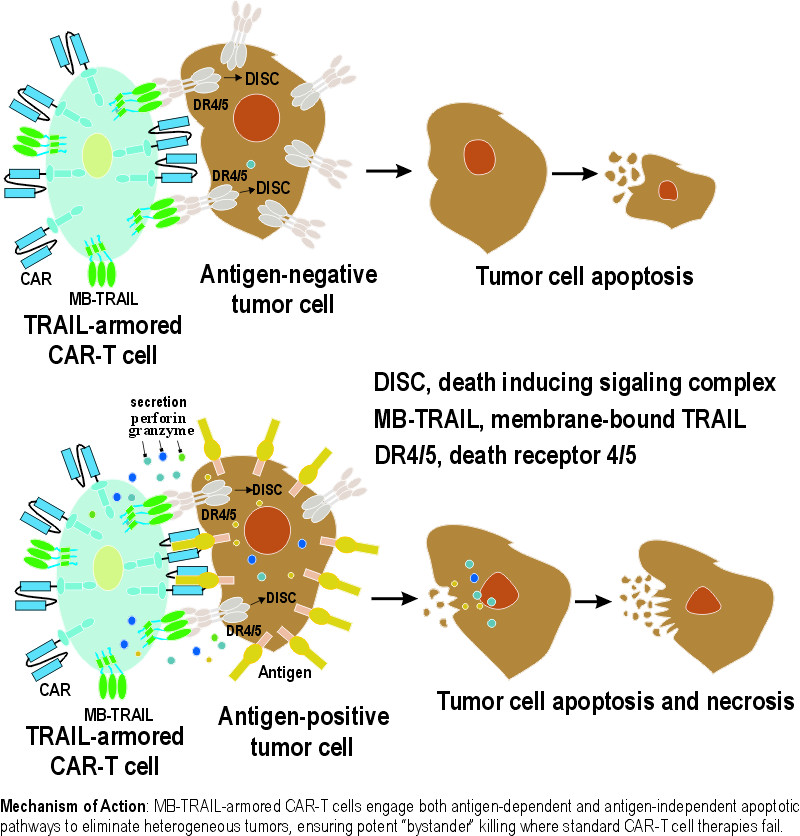
PrecisionAxis’ enhanced TRAIL-based fusion proteins deliver 100x the apoptotic signal compared to previous clinical versions of TRAIL, ensuring death receptor activation in CAR-resistant malignancies.
In vivo studies demonstrate sustained tumor eradication in 40% and 20% of mice challenged with heterogeneous non-small cell lung cancer (NSCLC) and pleural mesothelioma (PM) tumor cells, respectively. By providing a secondary, antigen-independent apoptotic signal, PrecisionAxis’ platform eliminates the antigen-negative 'escape' variants that typically lead to relapse in conventional CAR-T cell therapies.
By leveraging potent TRAIL-mediated apoptotic pathway, PrecisionAxis’ armored T cells eliminate neighboring tumor cells that lack the primary CAR-specific antigen, solving the problem of tumor heterogeneity
PrecisionAxis’ platform achieves a 10,000-fold reduction in the effective TRAIL concentration needed for tumor clearance. This shift dramatically expands the therapeutic window, ensuring potent apoptotic induction without the risk of systemic toxicity.
Beyond oncology, PrecisionAxis’ platform is being engineered to modulate immune responses in autoimmune pathologies. Our TRAIL-integrated approach holds significant potential for treating Type 1 Diabetes (T1D), Inflammatory Bowel Disease (IBD), and other immune-mediated disorders.
PrecisionAxis’ is building a robust and innovative intellectual property portfolio focusing on our TRAIL-armored CAR-T cell platforms. We are actively seeking strategic partnerships and collaborations to accelerate our lead candidates through IND-enabling studies and move our platform into the clinic.
Advance the Future of Immunotherapy
PrecisionAxis is actively seeking strategic partnerships, academic collaborations, and investment to accelerate our TRAIL-armored CAR-T-cell platform toward clinical entry. Reach out to discuss our data or explore possible collaboration
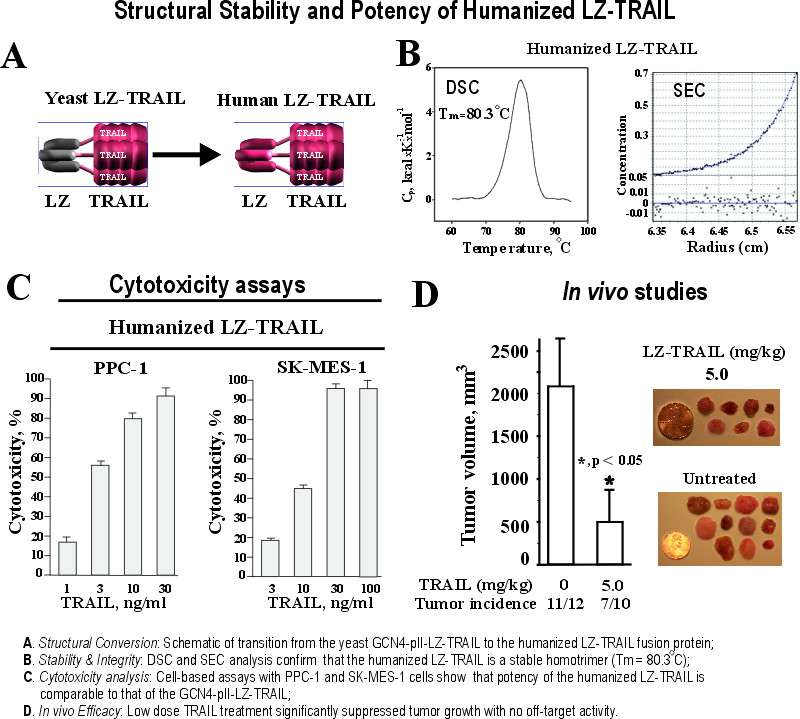
Engineering Leucine-Zipper-TRAIL: Augmenting Therapeutic Potency and Stability for Clinical Application
Achieving a 100-fold increase in cytotoxic potency and dramatically extended half-life in the bloodstream compared to clinical-grade TRAIL
PrecisionAxis’ technology overcomes the dual limitations that halted previous clinical TRAIL candidates: extreme structural instability and poor pharmacokinetics. By engineering a Leucine-Zipper (LZ)-TRAIL homotrimer, we successfully extended the therapeutic half-life to the 4–8 hour range, a 100-fold improvement over the 3.5-minute half-life of recombinant TRAIL. This optimized structural design yields a highly stable trimer with a melting temperature (Tm) of 81.1°C, as validated by SEC and DSC analysis.
This enhanced molecular stability translates to a 100-fold increase in cytotoxic potency across diverse cancer cell lines compared to clinical-stage TRAIL (dulanermin/TRAIL.0). In vivo, our platform achieved 75% tumor growth inhibition (TGI) at a low-dose (5 mg/kg) without the hepatotoxicity historically associated with modified death receptor ligands. This work establishes the high-potency, stable protein scaffold that serves as the foundation for our entire clinical immunotherapy pipeline.

To advance the Leucine-Zipper (LZ)-TRAIL platform toward clinical translation, we replaced the yeast-derived GCN4-pII trimerization motif with a proprietary human-derived LZ sequence. This Department of Defense (DoD)-funded research successfully minimized the risk of clinical immunogenicity without compromising the molecule's structural integrity or functional activity. Biophysical characterization via Differential Scanning Calorimetry (DSC) confirmed a melting temperature (Tm) of 80.3°C, ensuring high thermodynamic stability equivalent to our first-generation GCN4-pII-LZ-TRAIL construct.
Sedimentation equilibrium centrifugation (SEC) validated that the humanized LZ-TRAIL chimera maintains a stable homotrimeric state. Importantly, this humanized variant retains the 100-fold potency of the original GCN4-pII-LZ-TRAIL version, exhibiting equivalent efficacy in suppressing tumor growth in vivo. This conversion eliminates the potential for an adverse immune response while providing a stable, potent, and 'human-ready' therapeutic scaffold for our next-generation LZ-TRAIL-armored T cell therapies.
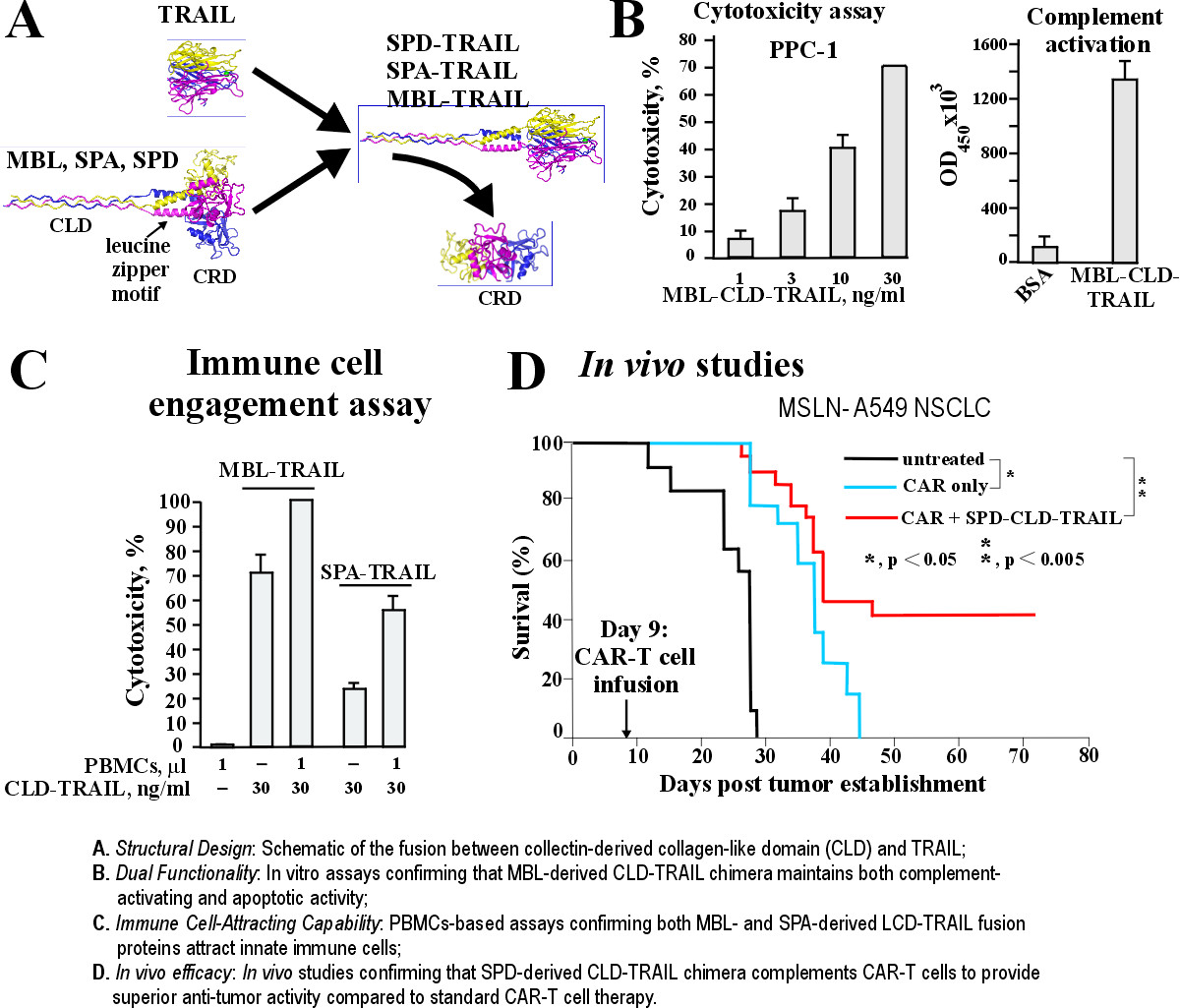
PrecisionAxis is expanding the therapeutic capability of the humanized LZ-TRAIL platform by incorporating collectin-derived collagen-like domains (CLD). These novel fusions, derived from Mannose-Binding Lectin (MBL) and Surfactant Proteins A and D (SPA and SPD), complement the dual-targeting activity of TRAIL-armored CAR-T cells by facilitating innate immune recruitment and complement activation.
Our data demonstrate that CLD-TRAIL chimeras retain high cytotoxic potency while successfully activating the complement system and recruiting phagocytes to the tumor site. By engaging innate immune effectors, we amplify the anti-tumor response to overcome antigen heterogeneity and mitigate the escape of TRAIL-resistant cell populations via enhanced bystander killing. In orthotopic lung cancer models, SPD-CLD-TRAIL demonstrated superior therapeutic activity over standard CAR-T cells, achieving sustained tumor eradication and a 40% long-term survival plateau without detectable systemic toxicity. This 'triple-threat' strategy provides a comprehensive solution to two critical barriers in solid tumor therapy: antigen loss and apoptotic resistance.
PrecisionAxis is advancing a modular, multi-factor immune platform designed to overcome the most hostile tumor microenvironments (TME). Our future pipeline focuses on the integration of intracellular signaling domains directly into the LZ-TRAIL scaffold, creating bi-functional molecules that trigger tumor apoptosis while simultaneously driving intrinsic T-cell activation.
Furthermore, we are optimizing multi-cistronic delivery systems to co-express a suite of auxiliary factors within our armored CAR-T cells. These include pro-survival cytokine receptors to enhance persistence, costimulatory ligands to recruit endogenous effectors, and chimeric 'switch' receptors designed to convert inhibitory signals (e.g., PD-1/TIGIT) into stimulatory ones. This comprehensive engineering approach ensures our armored CAR-T cells remain active, durable, and resistant to exhaustion in immunosuppressive environments.
A cornerstone of this platform is the CAR-mediated targeted delivery of our membrane-confined LZ-TRAIL. As described in our research currently in press at the Journal for ImmunoTherapy of Cancer (JITC), our armored CAR-T cells achieve equivalent therapeutic outcomes at a 10,000-fold lower TRAIL dose than soluble TRAIL treatment. This dramatically expands the therapeutic window, providing the essential safety foundation required to support complex, multi-factor armoring without systemic toxicity.
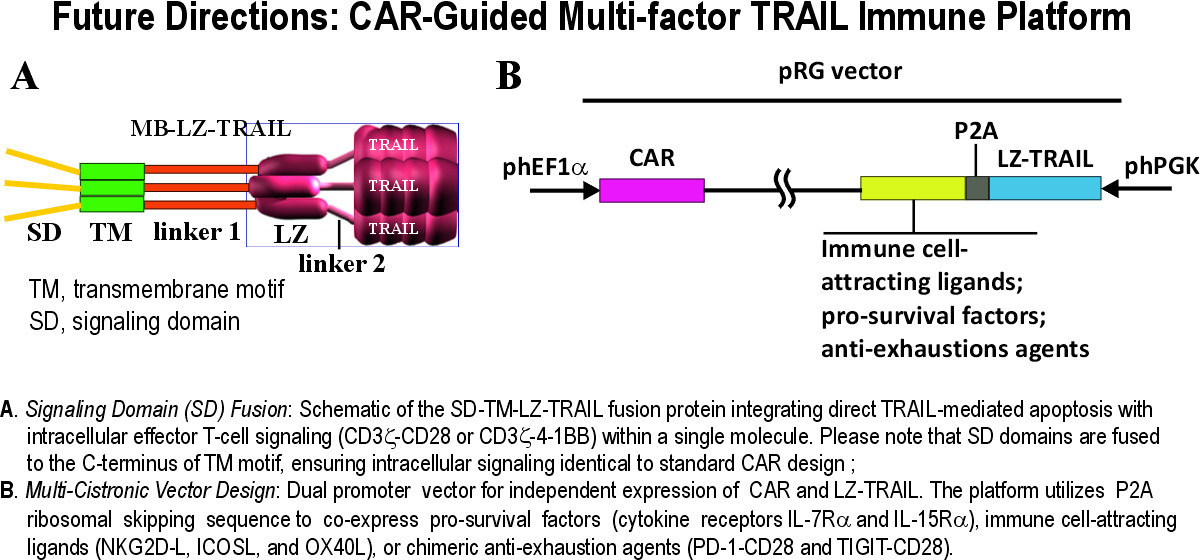
PrecisionAxis is expanding the capabilities of our stabilized LZ-TRAIL scaffold to address the root causes of chronic autoimmune and inflammatory disorders. While standard treatments often rely on broad, systemic immunosuppression, our platform enables a targeted 'Precision Immune Reset.' By armoring engineered T cells and Regulatory T cells (Tregs) with tissue-specific targeting mechanisms, we achieve a dual-action effect: the selective elimination of pathogenic activated immune cells and the simultaneous molecular protection of healthy tissues.
Leveraging the 10,000-fold dose-sparing advantage of our membrane-confined TRAIL design, we achieve superior therapeutic outcomes at significantly lower concentrations than soluble alternatives. This localized, high-impact expression, which can be further refined by inducible promoters, allows for an aggressive resetting of the immune microenvironment while maintaining a safety profile that far exceeds current clinical protocols. By concentrating the therapeutic load specifically at the site of inflammation, we maximize tissue repair and restore long-term immune homeostasis without the risks of systemic toxicity.
Frequently asked questions
Can't find an answer? Don't hesitate to reach out
What is PrecisionAxis Bio’s core mission?
PrecisionAxis Bio is a pre-clinical-stage biotechnology company dedicated to solving the most critical challenges in immunotherapy. Our mission is to advance cell therapy from simple antigen targeting to comprehensive, durable disease elimination. Whether we are treating solid tumors or autoimmune pathologies, our goal is to achieve complete regression and long-term immune balance through precision engineering and targeting.
What is the primary problem PrecisionAxis is solving?
In oncology, we are solving the "antigen escape" problem, where tumors hide from standard CAR-T cell therapies. In autoimmunity, we are solving the problem of broad immunosuppression by moving toward "Precision Immunomodulation" where we selectively remove disease-causing cells while simultaneously protect healthy tissue.
What are "Armored" CAR-T cells?
Standard CAR-T cells are often defeated by tumor heterogeneity where some tumor cells lack the target antigen. PrecisionAxis "armors" these cells by integrating them with our proprietary, stabilized TRAIL (TNF-Related Apoptosis-Inducing Ligand) fusion proteins. This creates a dual-action weapon that kills tumor cells via the antigen-dependent CAR-mediated receptor signaling and via the antigen-independent TRAIL-directed apoptotic pathway.
What is the TRAIL platform and why is it revolutionary?
While natural "wild-type" TRAIL is extremely unstable with a half-life of only 3.5 minutes in mouse bloodstream, we have engineered it into a stabilized "Leucine Zipper" (LZ)-TRAIL trimer. This structural design extends its half-life to several hours and increases potency at least 100-fold compared to previous clinical versions.
More importantly, our platform acts as a modular therapeutic scaffold. It is designed as an "all-in-one" system that can integrate:
Immune-cell attracting domains & ligands to recruit the body's natural immune response.
TCR-activating domains to boost T-cell engagement.
Pro-survival factors & anti-exhaustion agents to ensure the therapy remains active in hostile tumor environments.
This comprehensive engineering approach ensures that our therapies remain active, durable, and resistant to exhaustion even in the most hostile tumor environments.
What is the proof that this engineering works?
Our research, recently published in the Journal for ImmunoTherapy of Cancer (JITC), demonstrates that armored MB-LZ-TRAIL CAR-T cells achieve sustained tumor eradication in 40% and 20% of mice challenged with heterogeneous non-small cell lung cancer and pleural mesothelioma tumor cells, respectively. Critically, our engineered platform achieves these therapeutic outcomes at TRAIL dose four orders of magnitude lower (10,000-fold) than conventional soluble TRAIL treatments. By concentrating the therapeutic load directly at the tumor site, we dramatically expand the safety window for clinical application, providing a potent solution for CAR-resistant solid tumors without the systemic toxicity observed in earlier clinical attempts.
What is "Bystander Killing" and why does it matter?
In solid tumors, not every cell looks the same. A standard CAR-T cell will ignore a tumor cell if it lacks the specific antigen. Our armored CAR-T-cells use either membrane-bound or soluble LZ-TRAIL fusion proteins to engage "Death Receptors" (DR4/5) on those neighboring cells. This antigen-independent killing ensures that even "escape" variants are eliminated, preventing the relapses common in conventional CAR-T cell therapies.
How does your technology apply to autoimmune diseases?
We apply the same stabilized LZ-TRAIL scaffold used in our cancer research to address chronic inflammation. By arming T cells or Regulatory T cells (Tregs) with both LZ-TRAIL and disease-specific targeting, we achieve a "Precision Immune Reset." LZ-TRAIL selectively removes the activated, self-attacking immune cells while providing "tissue shielding" to protect vital organs like the pancreas, gut, or joints.
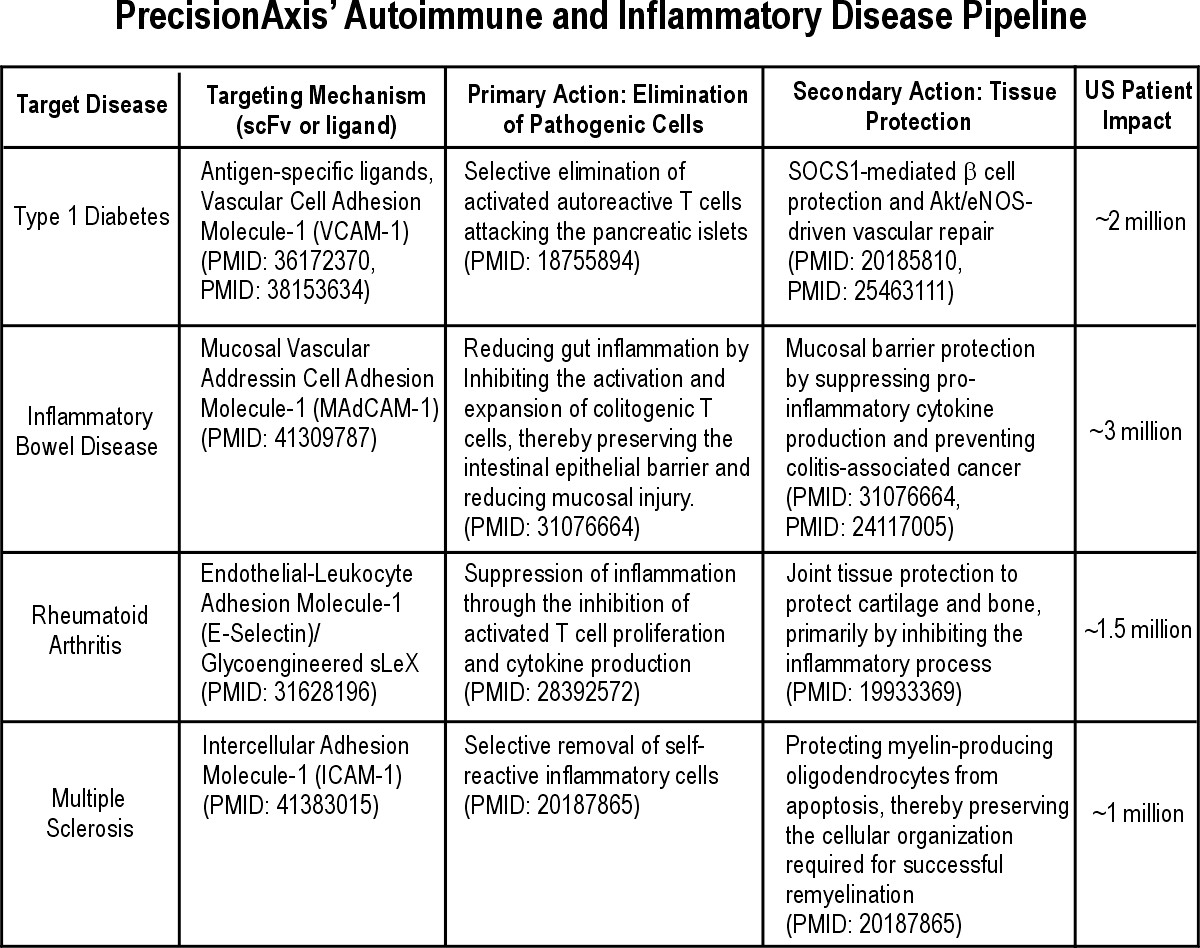
Which autoimmune diseases are you targeting?
We are targeting four major areas where our platform can provide a "dual-action" benefit of autoreactive immune cell removal and tissue protection, impacting approximately 7.5 million U.S. patients:
Type 1 Diabetes (2M): Protecting pancreatic Beta-cells.
Inflammatory Bowel Disease (3M): Preserving the intestinal barrier.
Rheumatoid Arthritis (1.5M): Protecting joint cartilage.
Multiple Sclerosis (1M): Protecting myelin-producing cells.
Is the platform safe for patients?
Yes. PrecisionAxis’ platform achieves a 10,000-fold reduction in the concentration of TRAIL needed for effective activity. This massive expansion of the therapeutic window ensures potent anti-tumor activity or immune cell induction without the systemic toxicity or liver damage historically associated with earlier versions of death receptor 4/5 (DR4/DR5) ligands. Additionally, our platform has been humanized to minimize the risk of an adverse immune response.
